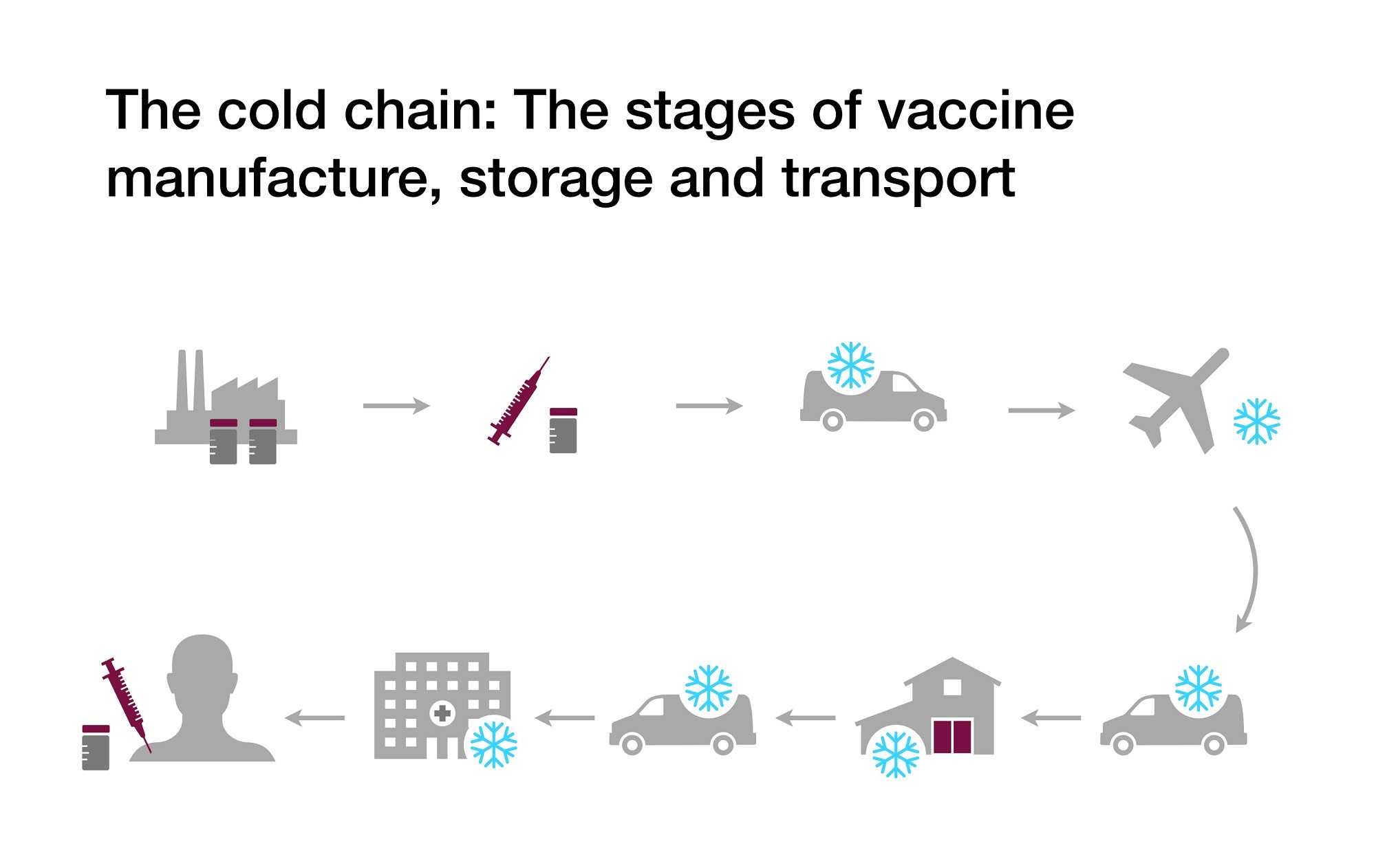
Vaccines have been an effective and economical way to prevent numerous diseases, and have saved millions of lives. However, in order for vaccines to remain effective, much care is needed. Vaccines in general are highly temperature sensitive products, and must be kept within the temperature ranges suggested by their manufacturers. If they are exposed to temperatures outside of these ranges, they can lose their potency, and can no longer be administered. As a result, the vaccines must be shipped, distributed, and stored within these temperatures from the time they are manufactured until the time they are injected. This process of transporting vaccines through a vast network of transporters, storage facilities and healthcare providers called the “cold chain”.
The cold chain is a very complex process with many possible pitfalls. As the containers are handed off from one point to the next, efficient procedures, accurate documentation, trained personnel and dependable equipment are all needed. Even one small delay or failure can have an affect on all parties involved, and can result in costly or even harmful consequences. At each step along the way careful adherence to CDC guidelines must be maintained, which involves properly packed shipments, regular temperature data logging, purpose built equipment, transport procedures and emergency planning.
The current pandemic has highlighted the importance of the “cold chain”. With the FDA’s emergency approval Pfizer’s new Covid-19 vaccine as of December 11, 2020 many new challenges have complicated this process even further. Pfizer’s new vaccine must be kept in a deep freeze of minus 94 degrees fahrenheit. While many of the cold storage facilities are already nearing capacity with existing medications, the volume of adding two doses for every person in the country will certainly cause crowding. And with the limited number of facilities with the ultralow freezers that Pfizer’s vaccine requires, the problem is likely to become compounded. Moderna, another large pharmaceutical company also has a vaccine that will likely be approved for emergency use, and will not require ultralow storage, but will still require two doses per person as well.
Along with the challenges of storage capacity, freezer types, logistics and accurate documentation, an added threat to the cold chain is the loss of power. Winter storms can cause frequent power outages causing cold storage devices to fail, resulting in the loss of entire vaccine inventories. This means that during a power outage, vaccines will often need to be moved from one location to another. However, this comes with its own challenges. Getting to these facilities during a storm with downed power lines and slippery roads can be dangerous. With the added size and cost of these inventories due to the Covid vaccines, generators and battery backup systems are needed to support these appliances, and will play an important role in maintaining the cold chain.



